PM Modi visit USA
PM Modi visit USA Only the mirror in my washroom and phone gallery see the crazy me : Sara Khan
Only the mirror in my washroom and phone gallery see the crazy me : Sara Khan Karnataka rain fury: Photos of flooded streets, uprooted trees
Karnataka rain fury: Photos of flooded streets, uprooted trees Cannes 2022: Deepika Padukone stuns at the French Riviera in Sabyasachi outfit
Cannes 2022: Deepika Padukone stuns at the French Riviera in Sabyasachi outfit Ranbir Kapoor And Alia Bhatt's Wedding Pics - Sealed With A Kiss
Ranbir Kapoor And Alia Bhatt's Wedding Pics - Sealed With A Kiss Oscars 2022: Every Academy Award Winner
Oscars 2022: Every Academy Award Winner Shane Warne (1969-2022): Australian cricket legend's life in pictures
Shane Warne (1969-2022): Australian cricket legend's life in pictures Photos: What Russia's invasion of Ukraine looks like on the ground
Photos: What Russia's invasion of Ukraine looks like on the ground Lata Mangeshkar (1929-2022): A pictorial tribute to the 'Nightingale of India'
Lata Mangeshkar (1929-2022): A pictorial tribute to the 'Nightingale of India' PM Modi unveils 216-feet tall Statue of Equality in Hyderabad (PHOTOS)
PM Modi unveils 216-feet tall Statue of Equality in Hyderabad (PHOTOS)India Open Competition in Shotgun, organised by the National Rifle Association of India (N
- Hockey India names Amir Ali-led 20-man team for Junior Asia Cup
- Harmanpreet Singh named FIH Player of the Year, PR Sreejesh gets best goalkeeper award
- World Boxing medallist Gaurav Bidhuri to flag off 'Delhi Against Drugs' movement on Nov 17
- U23 World Wrestling Championship: Chirag Chikkara wins gold as India end campaign with nine medals
- FIFA president Infantino confirms at least 9 African teams for the 2026 World Cup
Covid-19: DCGI approves phase II-III clinical trials of Covaxin on children aged 2 to 18 years Last Updated : 13 May 2021 01:04:29 PM IST 
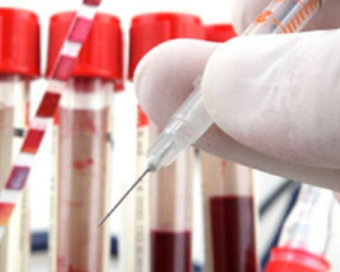
The Drugs Controller General of India (DCGI) has accorded permission to conduct the Phase II-III clinical trial of Covaxin (Covid vaccine) in the age group of two to 18-years-old to its manufacturer Bharat Biotech Ltd, Union Health Ministry said on Thursday.
"The trial will be conducted in 525 healthy volunteers," said the Ministry.
In the trial, the Ministry said, the vaccine will be given by intramuscular route in two doses at day 0 and day 28.
After careful examination, the DCGI accepted the recommendation of Subject Expert Committee (SEC). The national drug regulator of the country on Wednesday gave the approval to conduct the clinical trial in the age group of youngest age group till now paving way for them to be saved from the deadly pandemic outbreak.
Hyderabad-based Bharat Biotech International Ltd (BBIL) had proposed to carry out a Phase II-III clinical trial of Covaxin in the age group of two to 18 years.
As rapid regulatory response, the proposal was deliberated in Subject Expert Committee (SEC) (Covid-19) on May 11 this year.
The committee after detailed deliberation recommended for grant of permission to conduct proposed Phase II-III clinical trial to certain conditions.
IANS New Delhi For Latest Updates Please-
Join us on
Follow us on








172.31.16.186